Table of Contents (click to expand)
Anomers and epimers are both diastereomers, but an epimer is a stereoisomer that differs in configuration at any single stereogenic center, while an anomer is actually an epimer that differs in configuration at the acetal/hemiacetal carbon.
Before we talk about the differences between epimers and anomers in more detail, it helps if we first understand what stereoisomers are all about.
Recommended Video for you:
What Is A Stereoisomer?
In order to understand what a stereoisomer is, it’s crucial to know a thing or two about isomerism in general. An isomer is a molecule that has the same molecular formula as another molecule, but possesses different chemical properties. Another way to say this is that isomers contain the same number of atoms of each element, but the arrangement of these atoms is different.
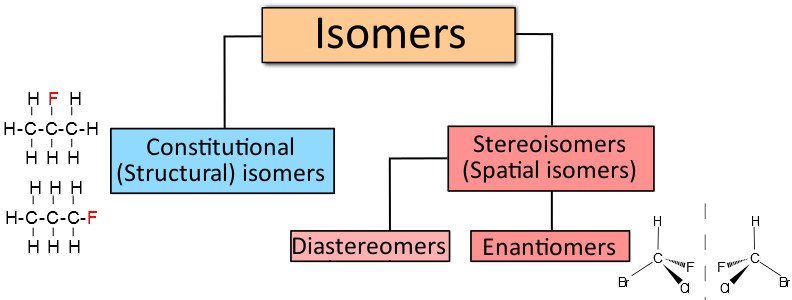
There are two kinds of isomerism; structural isomerism (wherein functional groups are attached in different ways) and stereoisomerism.
What Is Stereoisomerism?
Stereoisomers are isomeric molecules whose molecular formula is the same, but the 3-D orientations of their constituent atoms in space are different. Stereoisomers are further divided into two types: enantiomers and diastereomers.
Enantiomers are stereoisomers that are non-superimposable mirror images of each other. Also known as optical isomers, these have similar physical properties.

Diastereoisomers are those stereoisomers that have different configurations at one or more (but not all) stereoisomers without being mirror-images of each other. The term ‘epimer’ is used to refer to diastereomers that differ in configuration at only one chiral center.
An anomer is actually a kind of epimer that differs in configuration, especially at the acetal/hemiacetal carbon.
Epimer Vs Anomer
While an epimer is one of a pair of stereoisomers that differ in configuration at only one chiral (stereogenic) center, an anomer is actually an epimer (also a cyclic saccharide) that differs in configuration, especially at the acetal or hemiacetal carbon (refer to the image below to differentiate between acetal and hemiacetal carbons).
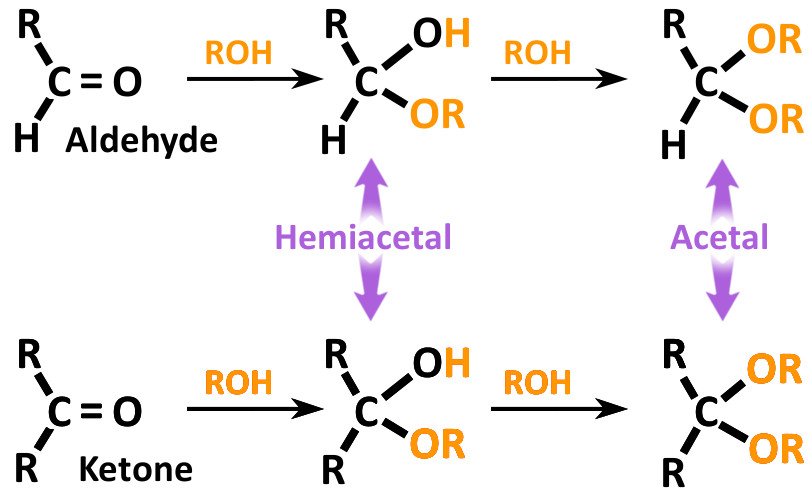
An epimer is one of a pair of stereoisomers that differ in configuration only at the chiral center. As such, all other stereocenters (if there are any) are the same in both molecules. If the pair of molecules has only 1 stereocenter, then the epimers are enantiomers, whereas when the molecules have 2 or more stereocenters, the epimers are referred to as diastereomers. (Source)
Refer to the following figure with two stereoisomers of chlorobutane.
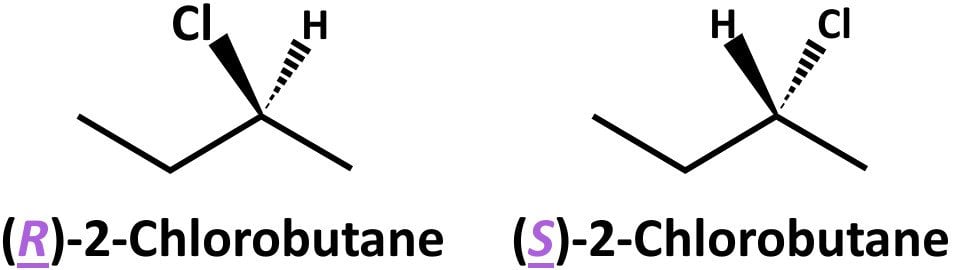
(R)-2-Chlorobutane and (S)-2-chlorobutane differ in absolute configuration at the C2 stereocenter. Note that 2-chlorobutane has only one stereocenter; that’s why these epimers are enantiomers.
On the other hand, in the stereoisomeric structures of tartaric acid, you can see that the two epimers (i.e., (2R,3R)-Tartaric acid and (2R,3S)-tartaric acid) differ in the absolute configuration at the C3 stereocenter.
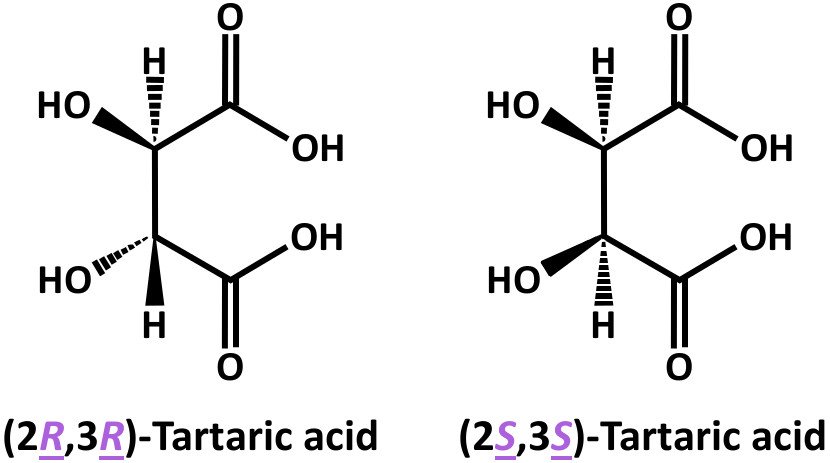
Note that tartaric acid has two stereocenters, which is why these epimers are diastereomers.
An anomer is a kind of stereoisomer; anomers are saccharides or glycosides that are epimers, which are distinct from each other in the configuration at C-2, if they are ketoses, or in the configuration of C-1, if they are aldoses.
In many cases, it so happens that carbohydrates exist in cyclic/acyclic forms. During cyclization, the carbon in the carbonyl group converts into a new stereocenter. Such cyclization results in the formation of two diastereomers, which differ in the position of the attachment of a certain functional group (Source). The new stereocenter is called the ‘anomeric carbon’.
The following figure should help to visualize this:
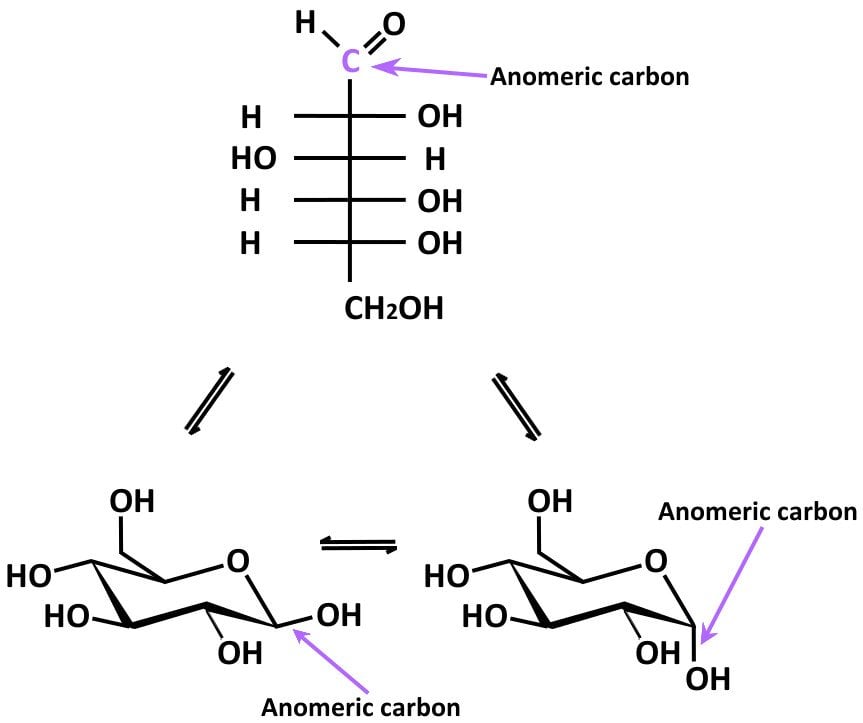
Two anomers are designated alpha and beta, depending on the configurational relationship between the anomeric center and the anomeric reference atom. If the hydroxyl group on C-1 and the -CH2OH group on C5 are on opposite sides of the six-membered ring, C1 is said to be the α anomer. If they are on the same side, C1 is said to be the β anomer.
Example: α-D-Glucopyranose and β-D-glucopyranose.
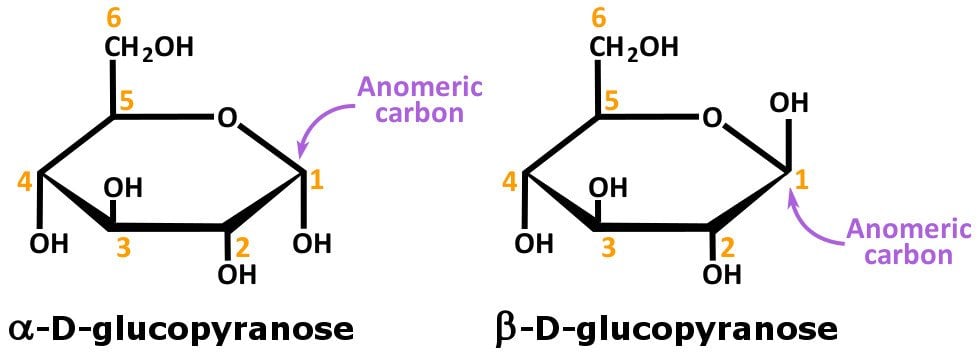
Note that the two stereoisomers (in the figure above) differ from each other in the configuration of C-1.
To conclude, both epimers and anomers are stereoisomers; in fact, anomers are a special case of epimers. The main difference between them is that epimers differ in configuration at only one chiral (stereogenic) center, but anomers differ in configuration, especially at the acetal or hemiacetal carbon.